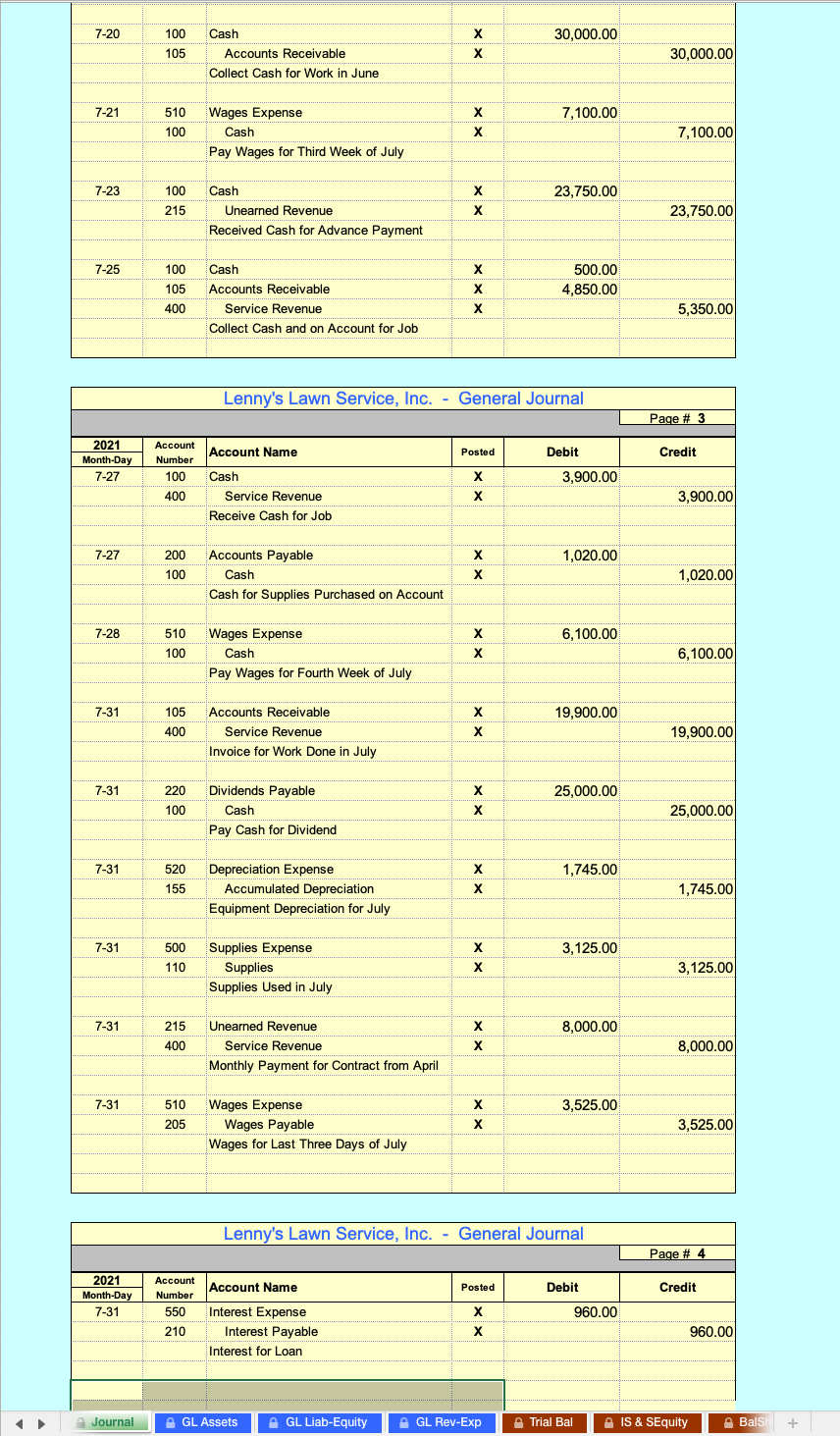
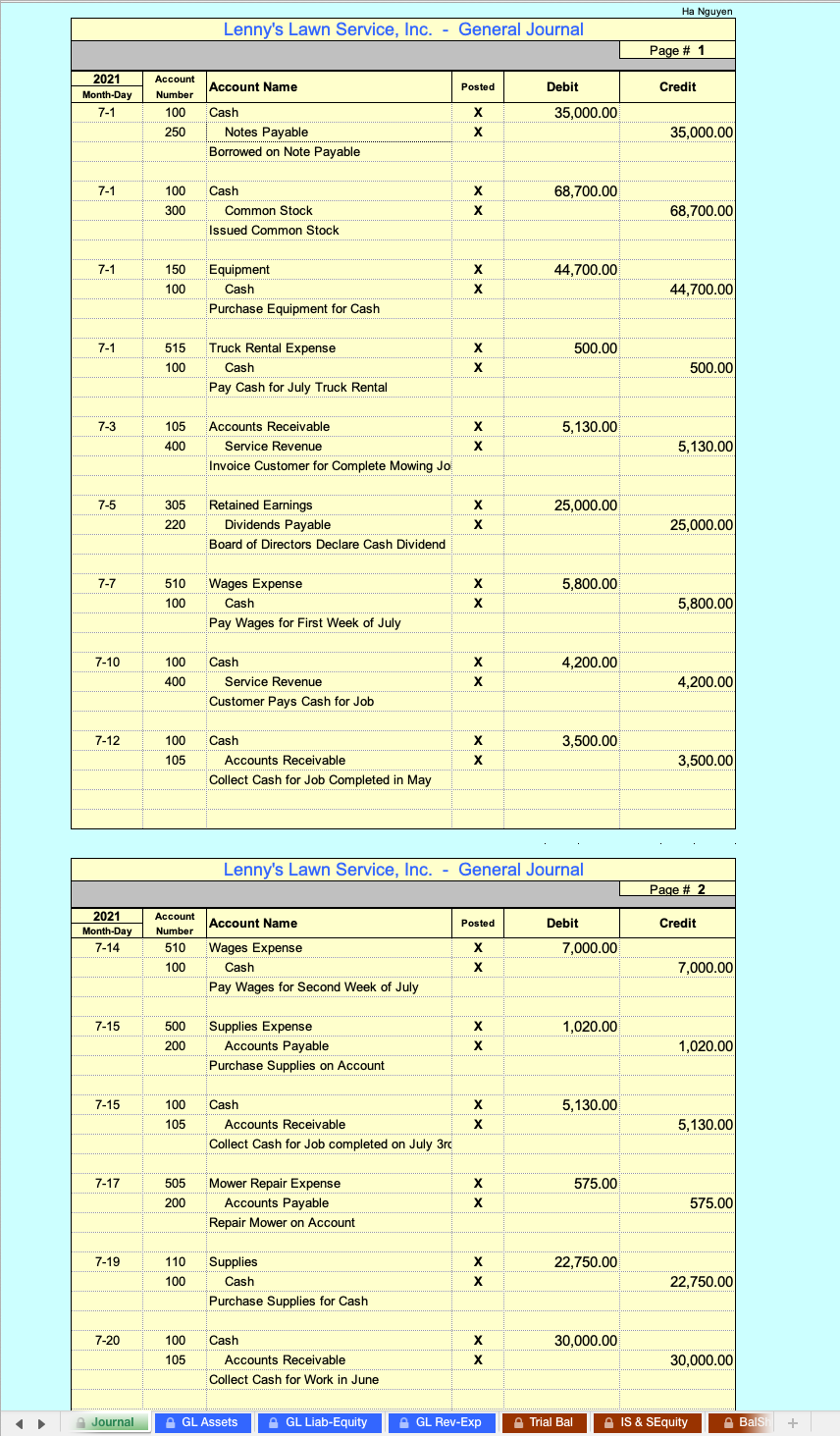
Cancers | Free Full-Text | Overcoming Challenges for CD3-Bispecific Antibody Therapy in Solid Tumors

Indian cancer doctors talk about the miracle drug 'Dostarlimab' trial that completely removed cancer: Do we have a cancer cure in sight? - Times of India

Second-line FOLFOX chemotherapy versus active symptom control for advanced biliary tract cancer (ABC-06): a phase 3, open-label, randomised, controlled trial - The Lancet Oncology

Safety and immunogenicity of the ChAdOx1 nCoV-19 (AZD1222) vaccine in children aged 6–17 years: a preliminary report of COV006, a phase 2 single-blind, randomised, controlled trial - The Lancet

Complex I inhibitor of oxidative phosphorylation in advanced solid tumors and acute myeloid leukemia: phase I trials | Nature Medicine

High-dose versus standard-dose twice-daily thoracic radiotherapy for patients with limited stage small-cell lung cancer: an open-label, randomised, phase 2 trial - The Lancet Oncology

Pictorial French Follies 3/1930-2nd issue-spicy stories, pix-art-pulp-VG/FN | Comic Collectibles - Magazines / HipComic

Aleix Prat #PrecisionOncology on Twitter: "Dr. Fatima Cardoso brings to imporant topics to discuss one day: 1) what should be the primary endpoint in advanced TNBC? OS? 2) implications for patients and

Effect of a 2-week interruption in methotrexate treatment versus continued treatment on COVID-19 booster vaccine immunity in adults with inflammatory conditions (VROOM study): a randomised, open label, superiority trial - The Lancet
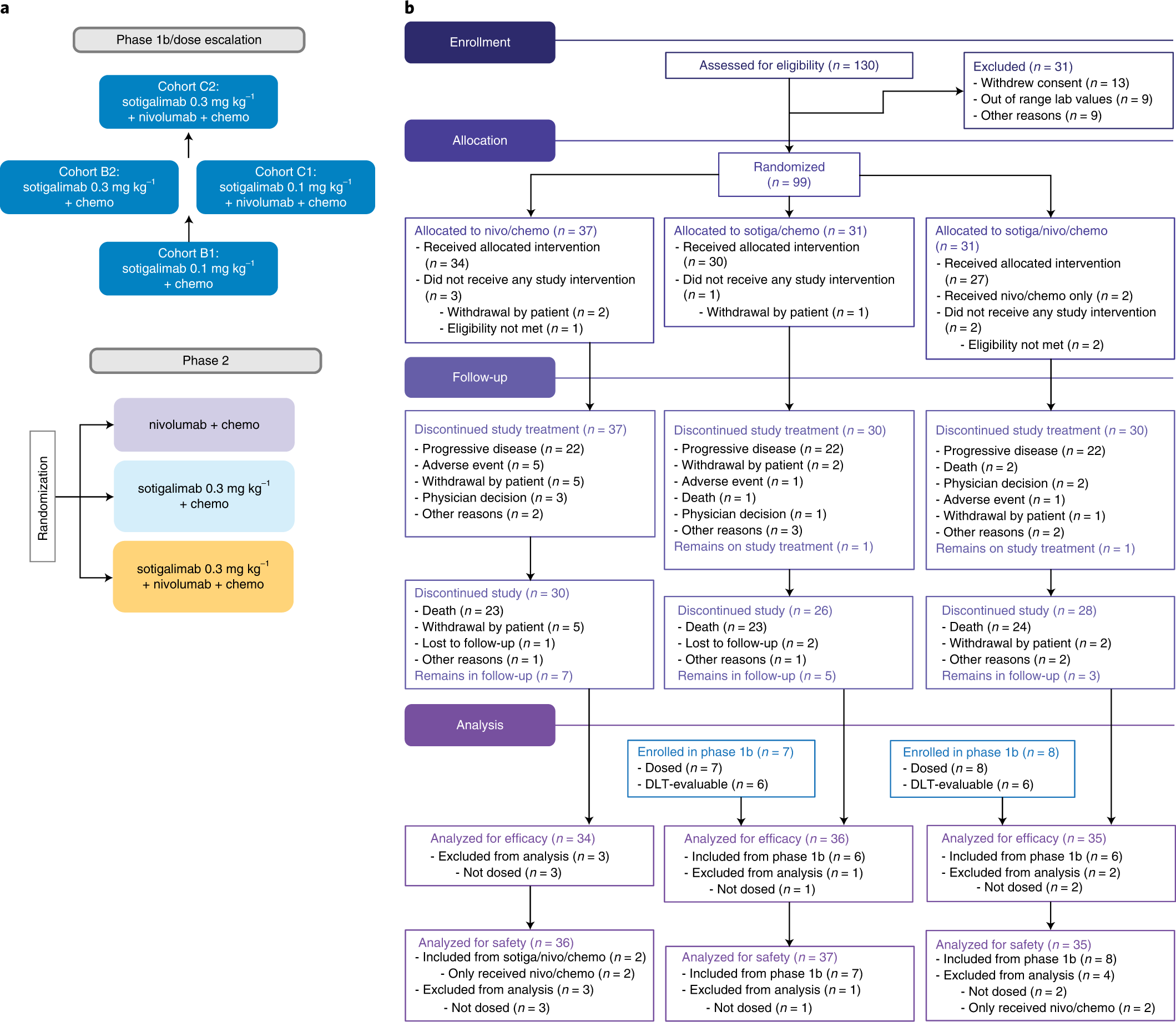
Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: clinical and immunologic analyses from the randomized phase 2 PRINCE trial | Nature Medicine